Your understanding of talc may still be limited to its role as a "filler."
As a plastics modification company, we cannot simply view talc as a "cheap filler to reduce costs." From a physicochemical and crystallographic perspective, talc is a functional mineral with unique microscopic geometry, complex surface chemistry, and nucleation effects. Below, I will move beyond the general notion of "increasing rigidity and reducing costs" and provide an in-depth analysis of the structural essence and modification mechanism of talc.
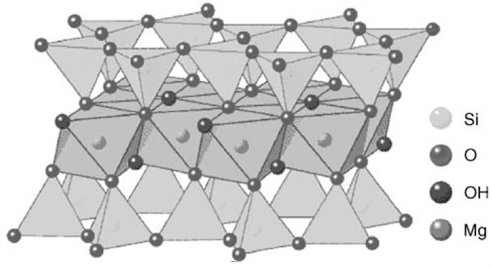
Crystal Structure and Interlayer Forces: Why Does It Possess Natural "Lubricity"?
The molecular-level "sandwich" structure of pure talc has the chemical formula Mg3Si4O10(OH)2. Crystallographically, it consists of two tetrahedral layers of siloxane (Si2O5) sandwiching a layer of brucite (Mg(OH)2) octahedral. This structure is chemically completely electrically neutral. (Mica flakes are charged)
Unlike mica (which relies on ionic bonds) or kaolin (which relies on hydrogen bonds), the interlayer forces in talc are held together by extremely weak van der Waals forces between adjacent "sandwich" layers.
The key role of interlayer forces: This weak force is the fundamental reason why talc is the softest mineral in nature (Mohs hardness 1). Under the high shear forces of plastic extrusion and injection molding, talc readily undergoes exfoliation (separation of flakes), which not only gives it solid lubricity, reducing wear on screws and molds, but also allows for the in-situ formation of microflakes with high aspect ratios (5:1 to 20:1) during processing.
2. Surface Groups and Chemical Properties: The "Huge Difference" Between the Basis and Edges
The surface chemistry of talc exhibits extremely strong anisotropy, which is crucial to the success of its surface modification:
The basal surface accounts for the vast majority of the total surface area. Because the exposed structure is a siloxane (Si-O-Si) network, lacking isolated polar groups, it cannot form hydrogen bonds with water. Therefore, the basal surface of talc is naturally hydrophobic and highly inert (while most minerals are hydrophilic).
The fracture edges occupy only a very small surface area. At the grinding fracture, extremely complex active sites are exposed, including: weakly acidic terminal silanol groups (HO-Si), weakly basic magnesium hydroxyl groups (Mg(OH)2), strongly acidic Brønsted sites, and Lewis acid sites. (These are very important.)
At pH values of 8.5–10.7, the crystal fracture edges expose weakly basic brucite structural sites, compounded by naturally occurring alkaline impurities such as magnesium carbonate in the ore.
The natural hydrophobicity of modified talc makes it easier to disperse in nonpolar polymers (such as PP) than other silicates. However, because its reactive hydroxyl groups are almost entirely confined to the edges, talc is "extremely insensitive" to traditional silane coupling agents (while silica and GF are extremely sensitive).




